|
Likewise, the positions of atoms in the molecule cannot change between two resonance contributors.ģ) All resonance contributors for a molecule or ion must have the same net charge.Ĥ) All resonance contributors must be drawn as proper Lewis structures, with correct formal charges. You can never shift the location of electrons in sigma bonds – if you show a sigma bond forming or breaking, you are showing a chemical reaction taking place (see rule #1). This means you cannot add, delete or move atoms around, even Hs!Ģ) Resonance contributors involve the ‘imaginary movement’ of pi-bonded electrons or of lone-pair electrons that are adjacent to ( i.e. In this text, carboxylate groups will usually be drawn showing only one resonance contributor for the sake of simplicity, but you should always keep in mind that the two C-O bonds are equal, and that the negative charge is delocalized to both oxygens.ġ) When you see two different resonance contributors, you are not seeing a chemical reaction! Rather, you are seeing the exact same molecule or ion depicted in two different ways. This is the kind of 3D picture that resonance contributors are used to approximate, and once you get some practice you should be able to quickly visualize overlapping 2 p z orbitals and delocalized pi electrons whenever you see resonance structures being used. Because there is one more electron than there are 2 p z orbitals, the system has an overall charge of –1. 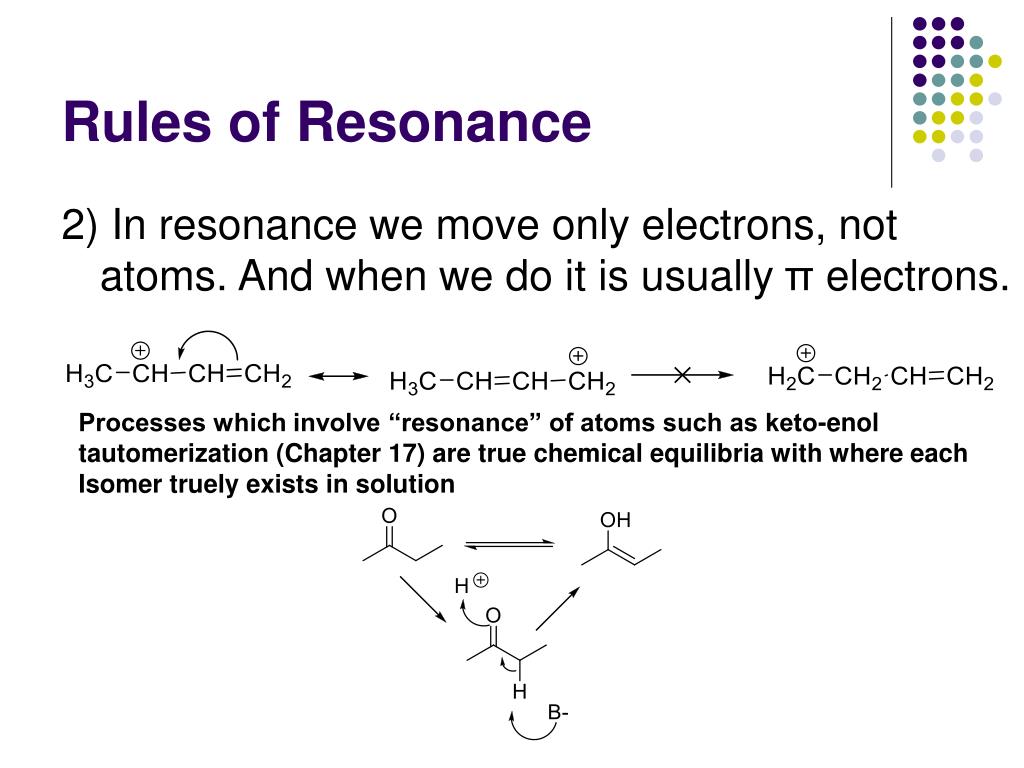
Overall, the situation is one of three parallel, overlapping 2p z orbitals sharing four delocalized pi electrons. Resonance contributor B, on the other hand, shows oxygen #2 participating in the pi bond with carbon, and oxygen #1 holding a lone pair in its 2 p z orbital. Resonance contributor A shows oxygen #1 sharing a pair of electrons with carbon in a pi bond, and oxygen #2 holding a lone pair of electrons in its 2 p z orbital. Rather, at all moments, the molecule is a combination, or resonance hybrid of both A and B. The depiction of benzene using the two resonance contributors A and B in the figure above does not imply that the molecule at one moment looks like structure A, then at the next moment shifts to look like structure B. Nevertheless, use of the curved arrow notation is an essential skill that you will need to develop in drawing resonance contributors. In the drawing of resonance contributors, however, this electron ‘movement’ occurs only in our minds, as we try to visualize delocalized pi bonds. A few chapters from now when we begin to study organic reactions – a process in which electron density shifts and covalent bonds between atoms break and form – this ‘curved arrow notation’ will become extremely important in depicting electron movement. 
Each of these arrows depicts the ‘movement’ of two pi electrons. In order to make it easier to visualize the difference between two resonance contributors, small, curved arrows are often used.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
